Nitrosyl azide
 | |
| Identifiers | |
|---|---|
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | N3−N=O |
| Molar mass | 72.027 g·mol−1 |
| Appearance | Pale yellow solid below −50 °C (−58 °F). Above that temperature it decomposes. |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Nitrosyl azide is an inorganic compound of nitrogen and oxygen with the chemical formula N3−N=O. It is a highly labile nitrogen oxide with the empirical formula N4O.
Synthesis
Nitrosyl azide can be synthesized via the following reaction of sodium azide and nitrosyl chloride at low temperatures:
Properties
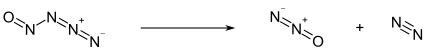
Below −50 °C, nitrosyl azide exists as a pale yellow solid. Above this temperature, it decomposes into nitrous oxide N2O and molecular nitrogen N2:[1]
Characterization of the compound with IR and Raman spectroscopy show absorption bands that agree well with calculated values for a trans-structure.[1][2] Quantum chemical calculations show a cis-form higher in energy by 4.2 kJ/mol and an aromatic ring form (oxatetrazole N4O) that is more stable by 205 kJ/mol. However, the cyclization to the ring form would have to surpass the 205 kJ/mol activation energy barrier require to bend the azide group, which might explain why nitrosyl azide is stable enough to be isolated at low temperature.[1]
References
- ^ a b c Schulz, Axel; Tornieporth-Oetting, Inis C.; Klapötke, Thomas M. (1993). "Nitrosyl Azide, N4O, an Intrinsically Unstable Oxide of Nitrogen". Angewandte Chemie International Edition in English. 32 (11): 1610–1612. doi:10.1002/anie.199316101.
- ^ Lucien, Harold W. (1958). "The Preparation and Properties of Nitrosyl Azide". Journal of the American Chemical Society. 80 (17): 4458–4460. doi:10.1021/ja01550a004.
- Cotton, F. Albert & Geoffrey Wilkinson (1999). Advanced Inorganic Chemistry (6th ed.). New York: John Wiley & Sons. p. 331. ISBN 0-471-19957-5.
- v
- t
- e
| HN3 | He | ||||||||||||||||||||
| LiN3 | Be(N3)2 | B(N3)3 | CH3N3 C(N3)4 CO(N3)2 | NH4N3 N3NO N(N3)3 H2N–N3 | O | FN3 | Ne | ||||||||||||||
| NaN3 | Mg(N3)2 | Al(N3)3 | Si(N3)4 | P | SO2(N3)2 | ClN3 | Ar | ||||||||||||||
| KN3 | Ca(N3)2 | Sc(N3)3 | Ti(N3)4 | VO(N3)3 | Cr(N3)3 CrO2(N3)2 | Mn(N3)2 | Fe(N3)2 Fe(N3)3 | Co(N3)2 Co(N3)3 | Ni(N3)2 | CuN3 Cu(N3)2 | Zn(N3)2 | Ga(N3)3 | Ge | As(N3)5 | Se(N3)4 | BrN3 | Kr | ||||
| RbN3 | Sr(N3)2 | Y(N3)3 | Zr(N3)4 | Nb | Mo | Tc | Ru(N3)63− | Rh(N3)63− | Pd(N3)2 | AgN3 | Cd(N3)2 | In | Sn | Sb(N3)5 | Te(N3)4 | IN3 | Xe(N3)2 | ||||
| CsN3 | Ba(N3)2 | * | Lu(N3)3 | Hf | Ta | W | Re | Os | Ir(N3)63− | Pt(N3)62− | Au(N3)4− | Hg2(N3)2 Hg(N3)2 | TlN3 | Pb(N3)2 | Bi(N3)3 | Po | At | Rn | |||
| Fr | Ra(N3)2 | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| * | La(N3)3 | Ce(N3)3 Ce(N3)4 | Pr | Nd | Pm | Sm(N3)3 | Eu(N3)2 Eu(N3)3 | Gd(N3)3 | Tb | Dy(N3)3 | Ho(N3)3 | Er | Tm | Yb(N3)3 | |||||||
| ** | Ac(N3)3 | Th(N3)4 | Pa | UO2(N3)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | |||||||